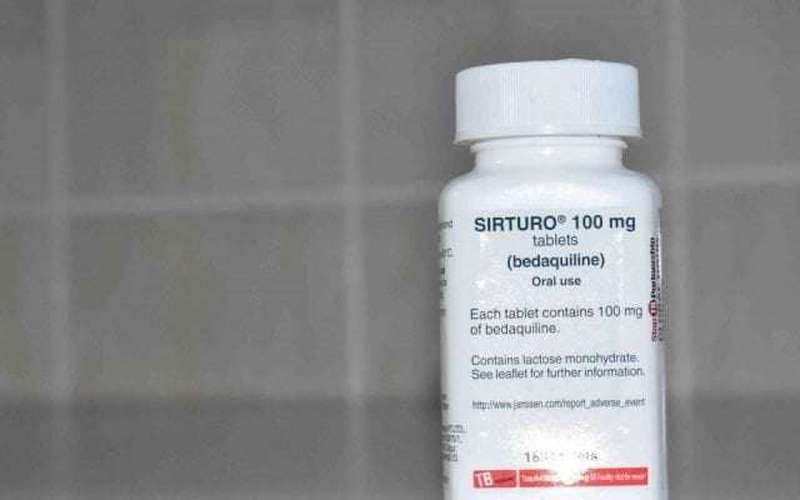
Bedaquiline is one of the most important components of the drug regimen prescribed for DR-TB patients.
Globally, there are 4,50,000 DR-TB patients, a vast majority of whom are in the LMICs, including India.
This development comes after decades of public pressure and efforts of TB advocates who have been demanding that the company must give up its call for what is called a ‘secondary patent’ – or the extension of its primary patent.
J&J’s patent over the base compound of the drug was set to expire in most countries in 2023.
However, it had applied for a secondary patent to extend what the activists termed a ‘monopoly’ over the drug.
India’s patent office had rejected J&J’s application for a secondary patent in April 2023.
The ‘Stop TB Partnership’, through its ‘Global Drug Facility (GDF)’, supplies DR-TB drugs to several countries, or fulfils a part of their requirements. »