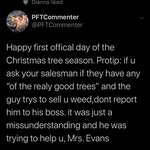
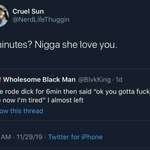
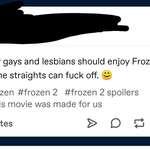
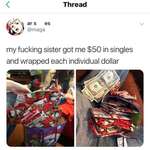
Researchers at Australia's Deakin University say they've managed to use common industrial polymers to create solid electrolytes, opening the door to double-density solid state lithium batteries that won't explode or catch fire if they overheat.
The new technology uses a solid polymer material, weakly bonded to the lithium-ion, to replace the volatile liquid solvents typically used as electrolytes in current battery cells.
The liquid electrolyte is the part of the system that becomes flammable during the kinds of infamous battery fires Samsung would rather forget.
In addition to making batteries safer, the team believes this solid polymer electrolyte will finally allow batteries to work with a lithium metal anode.
Getting that up toward 500 Wh/kg would allow massively extended ranges, or smaller, cheaper and vastly lighter battery packs.
It's not the 10X jump everyone seems to believe is coming down the pipeline, but it would be very significant.
The Deakin team says it has only used existing commercial polymers in the new process, meaning that industrial production should proceed with "little difficulty." »