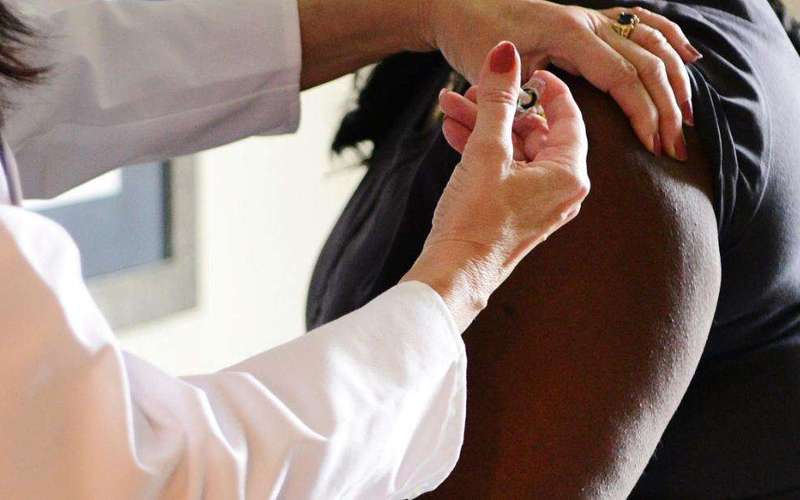
(CNN) Drugmaker Johnson & Johnson said Monday it has paused the advanced clinical trial of its experimental coronavirus vaccine because of an unexplained illness in one of the volunteers.
ENSEMBLE is the name of the study.
"Adverse events -- illnesses, accidents, etc. -- even those that are serious, are an expected part of any clinical study, especially large studies."
Johnson & Johnson's Janssen vaccine arm is developing the shot.
The company did not say what the unexplained illness was, but one point of clinical trials is to find out if vaccines cause dangerous side effects.
Trials are stopped when they pop up while doctors check to see if the illness can be linked to the vaccine or is a coincidence.
"Based on our strong commitment to safety, all clinical studies conducted by the Janssen Pharmaceutical Companies of Johnson & Johnson have prespecified guidelines. »