We did a dose-escalation, single-centre, open-label, non-randomised, phase 1 trial of an Ad5 vectored COVID-19 vaccine in Wuhan, China.
All of these vaccine platforms have advantages and disadvantages, and it is too soon to predict which will be more successful.
Our study suggests that there is potential for further investigation of the Ad5 vectored COVID-19 vaccine for prevention of COVID-19.
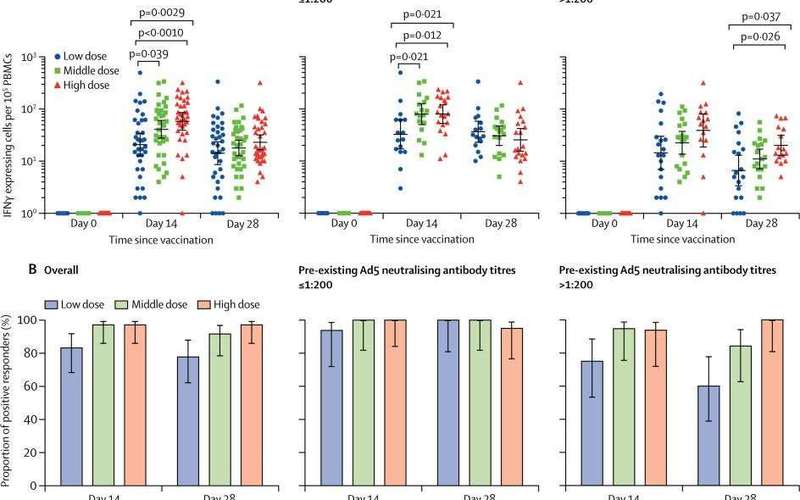
This first-in-human trial showed that the Ad5 vectored COVID-19 vaccine was tolerable and immunogenic in healthy adults.
Severe fever, fatigue, dyspnoea, muscle pain, and joint pain were reported in some of the recipients in the high dose group.
We assessed the number and proportion of participants with adverse reactions post-vaccination and compared safety profiles across the dose groups.
Multivariable analysis was used to establish the possible effects on the immunogenicity and safety profile of the vaccine candidates. »



![image for What's the biggest one hit wonder on Spotify? [OC]](6132cd5b-ab1a-50bd-b46b-9be0e1f5f9c4_thumb.jpg)






![image for A misty morning view of Yosemite Falls (CA) during sunrise (OC) [1333x2000] @ross_schram](eb1bb304-e332-52db-85e8-9faf0ef4851e_thumb.jpg)
