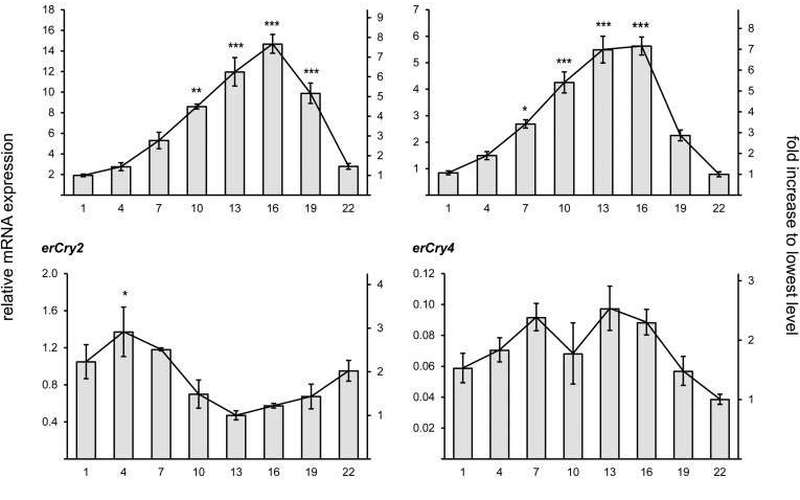
(A) The secondary structure of the erCry4 model obtained after extensive MD equilibration, with the isoalloxazine part of the FAD (red) and the conserved tryptophan triad (magenta).
(B) Superimposed molecular structures of the FAD isoalloxazine part and the tryptophan triad from atCry1 (blue), tetrad from dmCry1 (orange), and the tetrad from erCry4 model (red).
To include the complete structure of atCry1 and erCry4 would change the C-terminal part, which, unfortunately, cannot be predicted reliably yet.
(D and E) Comparison of the amino acid residues surrounding the FAD in the binding pocket of atCry1 (D) and dmCry1 and erCry4 (D and E).
The stability of the homology model can be established and probed by MD simulations, as done here for the model of erCry4.
(F) shows the root-mean-square deviation (RMSD) of the protein backbone atoms computed relatively to the initial structure of the model for erCry4 during the entire 0.5-μs MD simulation performed on our erCry4 model.
The gray area indicates the equilibration interval, while the remaining simulation was used to justify the stability of the model (production simulation). »